Discover the Wisdom of Nature
Welcome
Discover the Wisdom of Plants and Trees
The journey of Methow Valley Herbs began with a passion for plants and a vision to promote environmental awareness. Our founders recognized a growing need for trustworthy information about gardening, tree maintenance, and herbal healing in an era filled with misinformation. With a commitment to education, we have grown into a respected resource for nature lovers across the globe.

01
Medicinal Plants Education
Delve into the world of medicinal plants with our comprehensive guides. We provide researched information on herbs and their uses, along with tips for sustainable harvesting. Enhance your herbal wisdom and knowledge responsibly.
02
Tree Care Insights


03
Garden Care Guidance
Receive expert advice on garden care, including planting tips and seasonal recommendations. Our content empowers you to create a flourishing garden that is both beautiful and sustainable. Grow with confidence, every season.
Facilities and Amenities
Our Educational Offerings
Medicinal Plant Insights
Tree Care Guidance
Garden Care Resources
Our Blog
Tree Pruning Sydney: When Is The Best Time To Trim Trees
9 Things to Know Before Hiring Tree Removal Services Sydney
Mice Removal Sydney: Fast Solutions for Inner-City and Suburban Homes
What Is Included in Arborist Services Sydney for Residential Homes?
Pest and Termite Control: Why Combining Treatments Protects Your Property Long-Term
Choosing a Florist in Balgowlah Heights for Special Moments
Why Locals Choose Fresh Flowers in Greenwich
Green Waste Pickup in Australia: Sustainable Ways to Manage Garden and Tree Waste
Tree Loppers Near Me: How to Find Safe, Responsible Tree Care Services in Australia
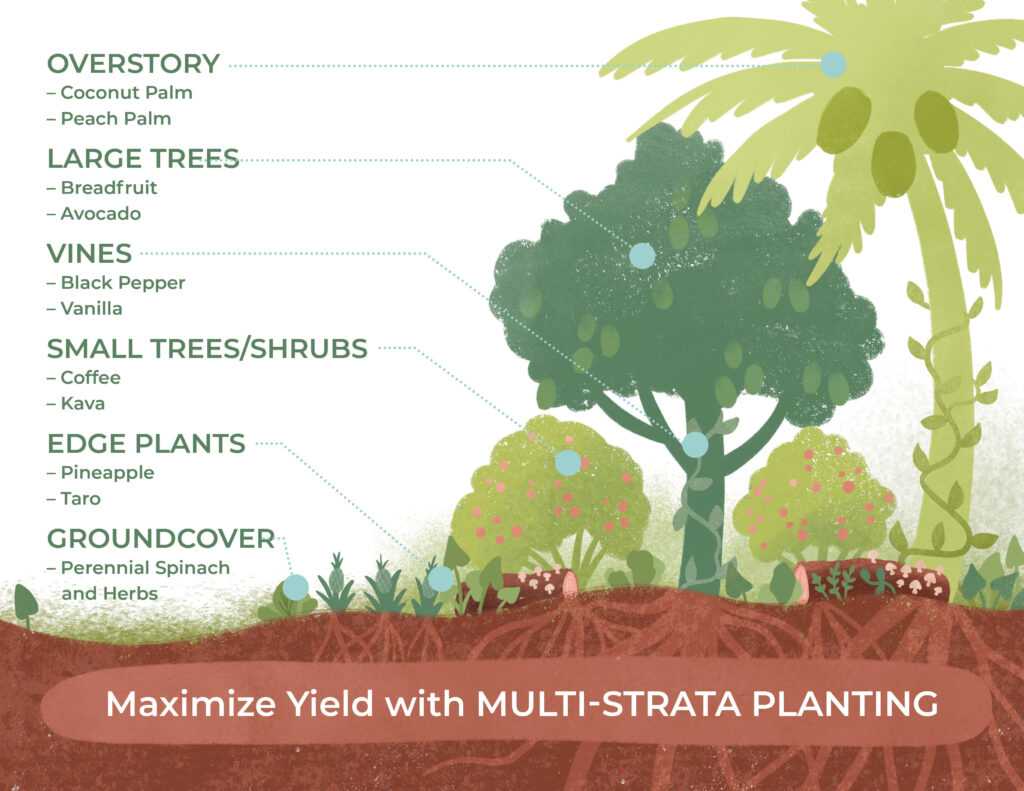
Guide to Agroforestry
Agroforestry is the modeling of our agricultural production after the forest ecosystem.
Kiss the Ground’s agroforestry guide explores the fundamental approaches and techniques used in agroforestry, highlighting how these methods are implemented within regenerative systems to strengthen soil vitality, improve farm resilience, and help reduce the impacts of climate change.
Testimonials
What Our Readers Are Saying
Methow Valley Herbs has been my go-to resource for understanding plants better. The information is always clear and trustworthy!
I appreciate the straightforward explanations and practical advice. This site has truly deepened my appreciation for nature.
An excellent platform for anyone looking to learn about gardening and herbs. I’ve found so much valuable information here.








